
These are almost chemically inert but form few compounds also.These can be liquified due to London dispersion forces.It means they have 8 electrons in their outer most shell and their electronic configuration is (ns 2, np 2) except He (1s 2) because their outer most shell contain only 2 electrons. Hence do not show tendency to gain or lose electron. These have very high ionization enthalpy and negligible electron affinity.are increases by increasing atomic number. These gases are odorless, colorless, tasteless with extremely low melting points and boiling points.Their atomic size increases by increasing atomic number due to increase in number of shells when we go from top to bottom in a group.They all are gases at room temperature.Radon is not found in atmosphere because it is radioactive element.And helium is present in the air while other noble gases are found in earth’s atmosphere. Helium is the most abundant element in the earth after hydrogen.The abundance of noble gases decreases as their atomic numbers increases.

Noble gases are found in atmosphere to extent about 1% by volume.The noble gas elements are found in the periodic table between the halogen elements and the alkali metals or last column of right side.So, let’s study about their occurrence, physical and chemical properties. To know more about it, we have to study about this topic in details like where are they found and what’re their physical and chemical properties, Then I hope you will understand about it properly. The atom size also affects the boiling point, This boiling point rises because the intermolecular forces between larger atoms with more electrons are greater than the force between smaller atoms with fewer electrons.When we moves down, the group number of electron shells increases, hence, the size of the atom will be increase.All the noble gases are colorless, odorless, tasteless and monoatomic that means they exist as single atoms.However, some noble gases can actually react to form certain compounds, that’s why this group is referred to as noble gases. They cannot not react with other elements therefore these are called the inert or unreactive gases.
#Printable periodic table of elements with noble gases full
All Nobel gases have full valence shells which makes them very stable elements.Helium has two valence shell electrons while each of the other noble gases has eight valence electrons.These elements having a full valence shell of electrons.
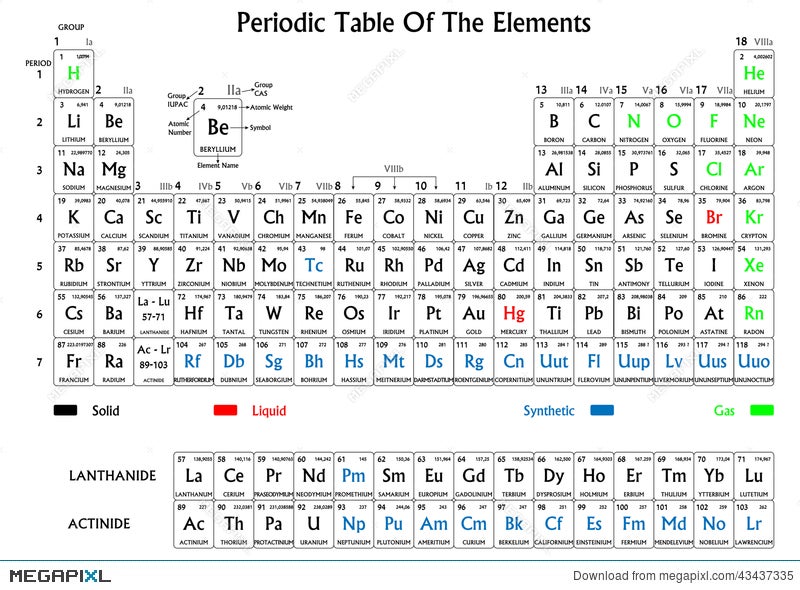
These elements include Helium (He), Neon (Ne), Argon (Ar), Krypton (Kr), Xenon (Xe), Radon (Rn) and Oganesson (Og). Noble gases are located as group 18 elements on the periodic table.

